Glioblastoma is one of the most formidable challenges in modern oncology—a cancer that often evades both conventional treatments and advanced immunotherapies. In the SelCell project, scientists at the Łukasiewicz – PORT are exploring a new strategy based on gamma-delta T lymphocytes. Their goal is to understand why immune cells from some donors can destroy tumor cells far more effectively than those from others.
The Challenge: Breaking Through the Tumor Microenvironment
Glioblastoma is a brain tumor whose location itself creates a barrier to many standard drugs. In addition, its tumor microenvironment is strongly immunosuppressive, effectively “switching off” the patient’s immune system. The statistics are stark: the five-year survival rate is only 5.1%.
One of the major hopes of modern oncology has been immunotherapy, including the breakthrough therapy based on CAR‑T cell therapy. Using genetic engineering, a patient’s own T lymphocytes are transformed into a sophisticated “living drug” capable of precisely recognizing and destroying cancer cells. While this approach has shown remarkable success in treating blood cancers—such as Leukemia and Lymphoma—its effectiveness against solid tumors remains limited. Glioblastoma belongs to this particularly difficult group.
Another obstacle is the therapy’s complexity: it requires isolating T cells from the patient, genetically modifying them, and manufacturing the treatment in a time-consuming and costly process.
For this reason, in the SelCell project conducted at Łukasiewicz – PORT, researchers are focusing on gamma-delta (γδ) T lymphocytes, which they believe may open the door to a new strategy for treating solid tumors.
Why Gamma-Delta (γδ) T Cells?
Gamma-delta (γδ) T cells make up only 1–10% of all lymphocytes circulating in human blood, yet they offer several important advantages over the cells used in CAR-T cell therapy.
“They do not require tissue compatibility, which means they do not have to come from the patient. They can be collected from a healthy donor and administered to an unrelated patient without the risk that they will attack healthy organs,” explains Dr. hab. Grzegorz Chodaczek, who leads the SelCell project and the Immunotherapy Research Group at Łukasiewicz – PORT. “Our lymphocytes are naturally cytotoxic. They recognize so-called stress antigens, which are specific proteins present only on the surface of cancer cells. By binding to them, they lead to their death – it is precisely in this mechanism that the therapeutic potential of gamma-delta T cells lies.”

In Search of the Ideal Donor
Earlier studies by the team showed that the cytotoxic activity of lymphocytes depends on the specific donor. In the project funded by the Medical Research Agency, researchers at Łukasiewicz – PORT aim to understand why cells from some individuals act as cancer “super-killers,” while those from others are far less effective.
Thanks to collaboration with the Regional Blood Donation and Blood Treatment Center in Wrocław, the team has access to biological material from healthy donors. From these samples, researchers isolate and expand two populations of gamma-delta (γδ) T lymphocytes, which are then tested against a panel of seven glioblastoma cell lines.
“We are looking for the molecular ‘signature’ of a donor and of lymphocyte cultures with the best anti-tumor properties. No one has yet explained which subtype of gamma-delta T cells is more effective or what type of receptor determines their ability to kill glioblastoma,” explains Dr. hab. Grzegorz Chodaczek, whose research group uses highly specialized instrumentation to search for answers to these questions.
Technology That Sees More
A unique technological process—the so-called pipeline—enables precise, multidimensional characterization of the process by which immune cells fight cancer. The research begins with large-scale cell culture in a dedicated incubator that provides identical and stable conditions for cells derived from multiple donors. This consistency is crucial for the reliability of the results.
The next stage involves deep phenotyping using advanced spectral cytometry. Unlike conventional methods, this technology allows researchers to analyze up to 40 markers simultaneously, providing an exceptionally detailed picture of cell condition, the presence of rare activation receptors, and signs of cellular exhaustion.
An integral part of the process is a high-throughput microscopy system that enables visualization of tumor cell killing in real time. Using this system, researchers can track the dynamics of the process, record the moment when glioblastoma cells die under the influence of lymphocytes, and—most importantly—generate quantitative data that allow comparisons of activity across cell cultures from different donors.
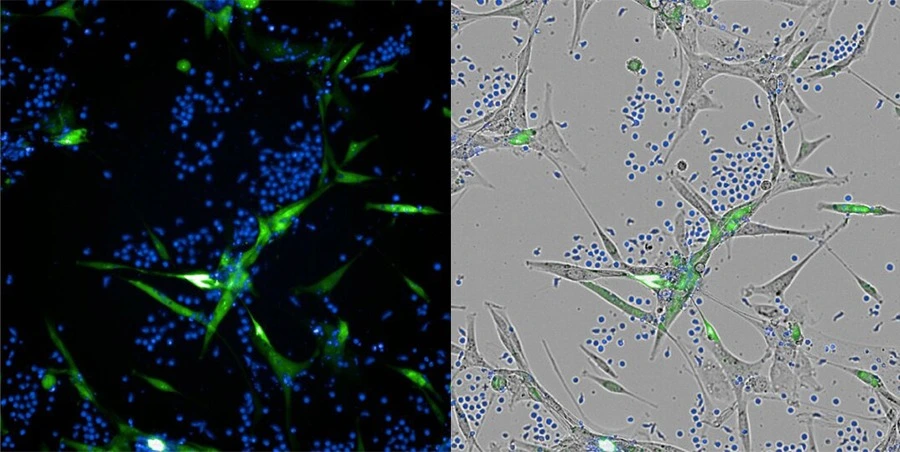
The workflow is complemented by transcriptomic and proteomic analyses using next-generation sequencing and mass spectrometry. These methods make it possible to identify the genes that become activated and the proteins produced by the most effective lymphocytes. This, in turn, helps researchers better understand the molecular basis of their effectiveness and identify new therapeutic targets.
“In immunotherapy for solid tumors, science is facing a wall that classical methods have not yet been able to break through. Thanks to such an advanced technological platform, we are not searching for new solutions blindly. We not only see which cells kill the tumor, but we are also beginning to understand why they do it,” adds Dr. hab. Grzegorz Chodaczek.
From Test Tube to a Patient Avatar
The goal of the project is to create a universal standard for selecting donors whose lymphocytes show the highest anti-tumor activity, paving the way for translating the biological potential of γδ T lymphocytes into an effective therapeutic strategy. Before this can happen, however, the Immunotherapy Research Group still faces several challenges that will be addressed in follow-up projects.
The next step will be to validate the findings in more advanced in vivo models. Researchers plan to reconstruct elements of the human immune system in model organisms in order to study interactions between lymphocytes and tumors in an environment that closely resembles the patient’s body. Such models will also make it possible to account for the highly specific, immunosuppressive tumor microenvironment.
The SelCell project therefore represents a crucial step in the development of an innovative therapeutic product based on gamma-delta T lymphocytes, bridging the gap between fundamental research and future clinical applications.




