For more than a century, science believed that neurons were responsible for our thoughts, emotions, and memory. The rest of the brain was treated as little more than filler. During Brain Awareness Week, we are telling the story of still little-known supporting players: astrocytes. Research shows that these star-shaped cells may hold the key to understanding depression and Alzheimer’s disease.
100 Years in the Shadow of Neurons
In 1856, German physician Rudolf Virchow described and named the substance between neurons “Nervenkitt,” or nerve glue – an insignificant filler. Forty years later, Hungarian anatomist Michael von Lenhossek examined these cells under the microscope and saw a star-like shape. He therefore called them astrocytes, from the Greek word “astron” (star).
For more than a century, scientific understanding of astrocytes changed very little: they maintain the proper chemical environment for neurons, regulate ion concentrations, and remove metabolic waste. Important and necessary, but not important enough to serve as a basis for scientific research.
It was not until the last decade of the XXth century that researchers discovered a communication between astrocytes and described that these cells are not passive observers, but active participant that “eavesdrops” on neuronal conversations and can actively modulate them. From that point, astrocytes began to attract attention, but the true revolution came only in recent years.
Breakthrough Years in Astrocyte Research
The years 2025 and 2026 have brought an unprecedented series of discoveries published in the world’s top scientific journals. Here are several that are reshaping neurobiology and the direction of future research.
Astrocyte: A Second Backup Copy of Memories
Until recently, memory was considered the exclusive domain of neurons. However, research by professor Inbal Goshen and Benjamin Deneen groups demonstrated that astrocytes form their own memory traces. During the learning process, a small subset of these cells (2–4%) activates alongside neurons, potentially creating a backup copy of our memories.
A study published in Nature in February 2026 goes even further, proving the vital role astrocytes in the amygdala play in forming and maintaining fear memory – a topic directly linked to the P4Health research area. The lead author of these discoveries, Dr. Andrew Holmes, presented their pioneering results at the PORT for Health: Neuroscience 2023 conference.
Norepinephrine: Concentration Needs an Intermediary
For decades, scientists believed that norepinephrine, the neurotransmitter responsible for alertness and concentration, acts directly on neurons. Recently, a series of studies published in Science overturned this dogma: norepinephrine acts on astrocytes first, and only then do astrocytes affect neurons. Fish whose astrocytes were “switched off” with a laser lost the ability to stop, because norepinephrine could no longer directly instruct neurons to do so. Studies of Dr. Michał Ślęzak, one of Łukasiewicz – PORT group leaders, showed that noradrenaline signaling to astrocytes is necessary for their capacity to listen to synapses.
CCN1 Protein: Controlling Brain Plasticity
A Nature paper revealed that CCN1, a protein produced by astrocytes, maintains the stability of connections in the adult brain. Without it, neurons lose their stable anchor points and begin forming random, short-lived links, leading to deterioration of regions such as the visual center. If science and medicine learned to control CCN1 levels, it might become possible to restore child-like plasticity to the adult brain. This, in turn, would open entirely new possibilities for rehabilitation after neurologic injury or stroke.
Astrocytic Leaf Architecture: Up to One Million Connections
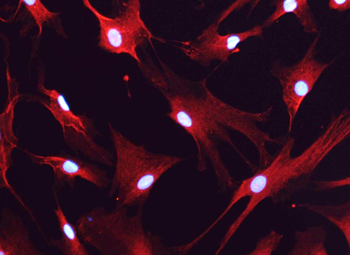
A publication in Cell explained the architectural power of astrocytes. Each astrocyte has thousands of microscopic protrusions (so-called leaves) that are 400 times thinner than a human hair.
A single astrocyte can wrap up to one million synapses at once. It acts like a processor, collecting distributed signals from thousands of neurons and organizing them into coherent information.
The Role of Astrocytes in Depression
The paradigm shift in thinking about the brain is especially visible in research on mental illness. A team led by Dr. Michał Ślęzak showed that disturbances in astrocyte function, not only neuronal function, may underlie depression.
These cells are the main recipients of stress hormones (glucocorticoids) in our nervous system and are the first to respond to adverse experiences in the body. When we experience chronic stress, astrocytic metabolic processes become impaired, causing abnormal neuronal operations in brain regions responsible for emotion regulation.
Research conducted in collaboration with big pharma industry confirmed that a specific astrocytic genetic profile was the most important feature distinguishing brain samples from individuals who died from suicide from samples from healthy controls. The problem is serious because key energy pathways and production of the most important neurotransmitters converge in astrocytes . Glutamate and GABA serve as the main accelerator and brake of neural signaling, so their balance underlies virtually every brain function, from mood stability to the ability to concentrate.
“Chronic stress disrupts this delicate mechanism. It creates chaos in communication between nerve cells and glia, and interrupting this dialogue leads to changes in molecular program of both partners. Hence, understanding astrocyte malfunction is crucial to provide better treatments of depression, which treatment remains a medical challenge,” says Dr. Ślęzak, the leader of the Biology of Astrocytes Research Group, who focuses on developing methods to normalize astrocyte metabolism. Restoring balance in the diseased brain would open the way to effective therapies for millions of people for whom currently available medications bring no improvement.
Astrocytes and Neurodegeneration
One of the critical functions of cells is maintaining so-called protein quality control, the system that detects and removes misfolded proteins. In a healthy brain, defective proteins are continuously repaired or removed. When they start to accumulate, they form toxic deposits characteristic of neurodegenerative diseases.
Dr. Agnieszka Krzyżosiak and colleagues hypothesized that targeting protein quality control could be exploited for therapeutic benefit and demonstrated the effectiveness targeting these “defense systems” against neurodegenerative phenotypes. The method they discovered for pharmacologically strengthening the cellular stress response produced results promising enough to lead to clinical trials in patients with ALS.
Although neurodegeneration is a disease of neurons, it is now becoming clear that astrocytes are active contributors to the pathology. Yet, astrocytic protein quality control pathways remain largely unexplored.
Research by Dr. Agnieszka Krzyżosiak and her team at Łukasiewicz – PORT suggests that failure of these astrocytic cellular protective pathways may be a tipping point in disease progression. Scientists hypothesize that modulating astrocytic proteostasis may benefit neurons by protecting them from progressive degeneration. This opens possibilities for the identification of more specific and selective drug targets in astrocytes, an unexplored therapeutic space.
“Thanks to a technology that allows us to reprogram cells taken, for example, from a patient’s skin into nerve cells, we can create brain models that preserve the patient’s biological age. We can reproduce the condition of a specific adult individual in the laboratory,” explains the leader of the Neurodegeneration Mechanisms Research Group. “Only this level of precision offers hope for therapies that, instead of merely alleviating symptoms, can truly stop degeneration across entire neuronal networks by restoring astrocyte function.”
Astrocytes at the Center of P4Health
After decades of focusing exclusively on neurons, looking at the brain as a whole ecosystem opens entirely new possibilities in medicine. Scientists see glial cells as a new therapeutic space, with potential for drugs that, unlike current ones, will target astrocytes rather than neurons.
Both Dr. Ślęzak’s team and Dr. Krzyżosiak’s team conduct research within the P4Health Center. With the official launch of the neuroscience International Research Agendas (MAB) program at the end of February, P4Health became one of very few scientific centers in the world to place astrocytes at the center of its research and development program.
Thanks to an EUR 8 million grant from the Foundation for Polish Science, the Wroclaw-based unit gained key tools to implement this vision. The project’s next phase assumes that additional scientists and new research groups will soon join the institute.
The entire MAB project, and one of the newly created groups, is headed by Prof. Bastian Hengerer. His team will focus on developing translational biomarkers and using innovative functional ultrasonography (fUS) to observe and measure how drugs or genetic modifications targeted directly at astrocytes can repair disrupted connections in neuronal brain networks.
The synergy of expertise across all four group leaders will enable researchers at P4Health to study astrocytes from every angle: from gene expression in single cells, through complex neuronal circuits, to preclinical validation of new treatment strategies.
However, this must be emphasized: these are still research directions, not ready-to-use therapies. Identifying glial dysfunction as a source of brain disease is only the beginning. A key stage in translating these discoveries into clinical practice will be experimental work aimed at validating therapeutic strategies that restore the physiologic function of astrocytes in models that enable objective biological measurements.